The 1st law of thermodynamics
|
The first law of thermodynamics states that energy can't be created and/or destroyed, only transformed. This is similar to the law of conservation of mass in that you can just make ice-cream or rabbits appear out of thin air; the first law of thermodynamics basically states that you can't just summon lightning bolts from your hands and shoot people you don't like. Instead energy has to be transferred from one place to another or already be there and be used in work. |
the 2nd law of thermodynamics
|
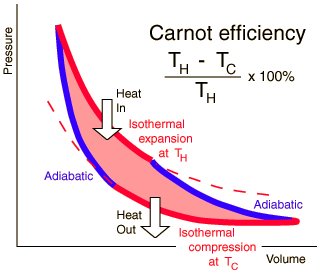
The second law of thermodynamics has several laws or statements that constrain the direction of heat transfer and the highest attainable efficiency of a heat engine, something that produces motive power from heat. 1. Heat will not spontaneously flow from a cold object to a hot object 2. A system within an adiabatic environment will become more disordered over time. This quantity can be determined in terms of something called entropy 3. It's impossible to create a heat engine that extracts all the heat of something and converts all of it into useful work. These laws restrict what heat engines and heat pumps can do. Without external work or an external force, heat cannot be directly from a cold reservoir to a warm place. Over time molecules will become misplaced due to atoms bouncing around. This is measured in entropy. As time moves on and the atoms are allowed to bounce around more, the state of the place will become more disordered. Entropy can also stand for the amount of energy that is unavailable to do work. Because it's impossible to create a one hundred percent efficient engine because heat cannot be transferred or moved without work done. |
|